
Health Canada Cannabis Packaging & Labeling Guidelines for LP’s
When planning your packaging solutions for your company, you should be aware of the guidelines required for your packaging and labelling. For Health Canada Licensed Producers, there are specific guidelines for what type of packaging you can use, what can and cannot be displayed on labels, as well as required product information, standardized warnings, and layout requirements for the information on the package display panels.
Contact us today for a consultation on how Dispensary Supply Canada can help your business get ready to hit the shelves! We offer graphic design services for all customers, and specialized design services for Health Canada Complaint packaging and labeling needs.

The Cannabis Regulations lay out requirements pertaining to how cannabis and cannabis products must be packaged and labeled prior to sale, distribution or export.
REGULATIONS REQUIRE:
1. Plain packaging and labeling for all cannabis products.
2. Restrictions on logos, colours, and branding.
3. Cannabis products must be packaged in a child-resistant container and be labeled with the standardized cannabis symbol, the mandatory health warning message, and include specific product information (e.g., brand name of the cannabis product, class of cannabis, delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) information, license holder information).
These requirements are set to reduce the risks of accidental consumption and overconsumption as well as reduce the appeal of cannabis products to young people. The required information provides consumers with the information they need to make informed decisions before using cannabis.
Licence holders are responsible for complying with the Act and Regulations, and other legislation that may apply to them or their activities. Check the federal and provincial guidelines to ensure you are operating within the regulations provided.
Health Canada does not review or pre-approve packages and labels of cannabis products, it is up to the Licensed Processor to adhere to the guidelines.
The Compliance and enforcement policy for the Cannabis Act can be found on Health Canada’s website for more information.
PACKAGING COMPLIANCE RULES
1. Packaging must be a single uniform colour. No fluorescent colours, containers or wrappers. Colour of container must create contrast with the yellow colour of the standardized yellow warning label, and the red colour of the standardized red hexagonal cannabis symbol.
2. Packaging must have a smooth texture. No raised features, embossing, decorative ridges, engravings, etchings, etc. on cantor and coverings. This does not apply to features necessary for opening or closing the packaging. Raised features are permitted for features that assist persons that are visually impaired.
3. Packaging may not contain hidden features. No heat-activated inks, UV inks, and other features viewed with other technological means, unless used to prevent counterfeiting. See Health Canada’s website for specifics on permitted counterfeit prevention features.
4. No features that change surface area, including tags or fold-out panels. Fold-out panels are permitted as an exception for immediate small containers. See section 8.3 of the Health Canada Cannabis Packaging and Labeling Guidelines for more information on this exception.
5. Packaging must not emit any scent or sounds.
6. No cut-out windows. Products must be stored and sold in packaging that is totally opaque.
7. Branding elements such as patterns or images are not permitted on the plain packaging. Images are not permitted on the interior or exterior of the container, covering, or wrappers. Containers may display one rectangular barcode in black and white without any other image or design. Exterior primary display panel of the container must display the standardized cannabis symbol at at minimum 1.27cm x 1.27cm.
8. No inserts or leaflets are allowed within or attached to the container.
9. Packages cannot contain more than one class of cannabis.
10. Packaging must prevent contamination of cannabis and cannabis seeds. It must keep contents of the package dry.
11. Packaging must be child-resistant. Child-resistant features must meet the requirements set out in C.01.001(2) to (4) of the FDR
12. Packaging must have a security feature. This feature provides reasonable assurance to the consumer that the package has not been opened or tampered with prior to purchase. This could include a security seal, a heat-sealed package, a shrink band, a tamper-evident sticker seal, etc.
13. Packages must not contain the maximum amount of cannabis. For dried cannabis, this is no more than 30 grams of cannabis.
14. Products may not contain more than the maximum allowed THC quantity. Cannabis topical or extracts may not contain more than 1000mg of THC per package. Cannabis edibles may not contain more than 10mg THC per edible.
15. Cannabis extracts not in discreet units must have control measures. Containers should not contain more than 90ml of liquid cannabis extract. The extract should not be able to be easily poured or drunk. Containers containing liquid cannabis extracts should have an integrated dispensing mechanism, such as a pump, metered spray, or accurate-dose dropper. Integrated dispensing systems should not dispense more than 10mg of THC per activation.
16. The limitations on THC content must account for the product’s THCA content to convert to THC.
17. Packaging for cannabis and cannabis products must be constructed with food-grade materials. All materials coming into contact with the product must also be made with food-grade materials. Wrappers may only be used if they are required to maintain the quality or stability of the cannabis product. Wrappers must also be made of approved food-grade materials. Section 186(a)(i),(ii), and (v) to (vii) of the Safe Food for Canadians Regulations (SFCR) outline requirements for a package including to be clean, suitable for its intended use, and free from odours that might affect the food [122.2, CR].
18. For co-packing regulations visit the Health Canada Cannabis Packaging Guidelines Section 7.4
Disclaimer: This is a comprehensive summary of the Health Canada cannabis packaging guidelines, yet it is the responsibility of the company to ensure they are in compliance with the current and complete requirements for packaging and labeling their products. For the complete and current list of requirements visit Health Canada’s list of prohibitions, requirements, and regulations pertaining to packaging and labeling of cannabis products. See website:
https://www.canada.ca/en/health-canada/services/cannabis-regulations-licensed-producers/packaging-labelling-guide-cannabis-products/guide.html
LABELING COMPLIANCE RULES
1. Labels or directly printed product information must be displayed on the Primary Display Panel of the package. The panel must be visible under normal or customary conditions of purchase or use.
2. The Primary Display Panel (PDP) must display the Standardized THC Warning Symbol Logo (Red Hexagon) (Shown Below)
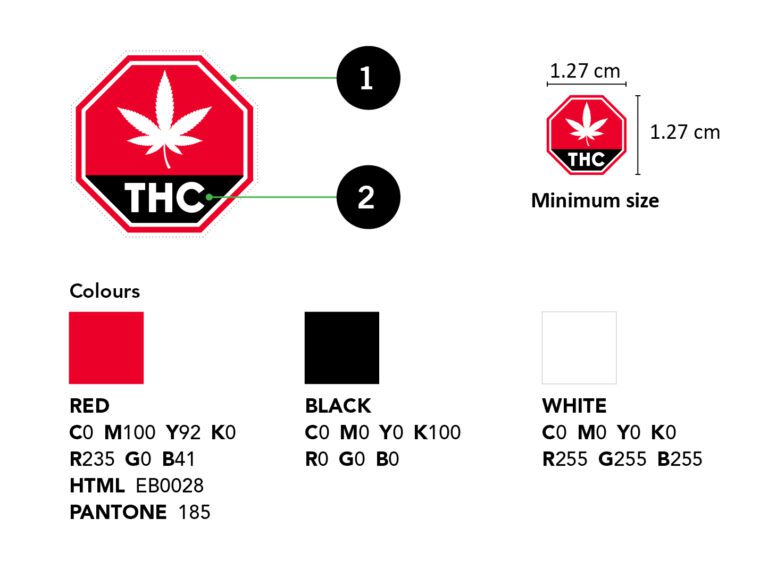
3. The PDP must display the standardized health warning message in a yellow box. (Shown Below)
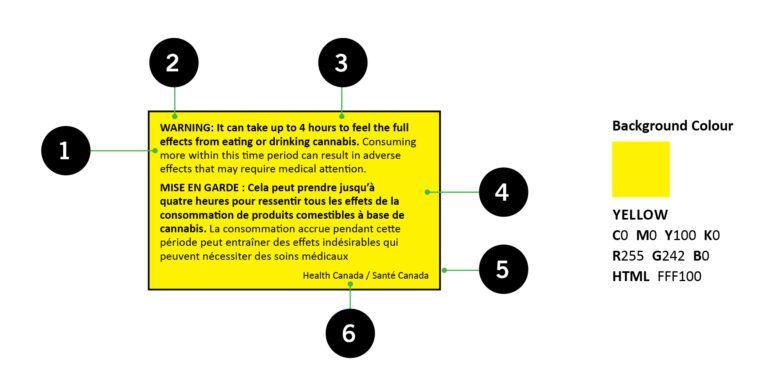
A list of approved stargazed warning messages can be found at this link: https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/laws-regulations/regulations-support-cannabis-act/health-warning-messages.html
4. The unit of measure of THC or CBD content must be represented by quantity (mg) or concentration (mg/g), depending on the class of cannabis. The intended use and whether or not the cannabis is in discreet units or not will determine how this information is displayed on the PDP.
5. The brand name of the cannabis product must be displayed on the PDP. Brand name text cannot be metallic or fluorescent colour and must be equal to or smaller than the size of the text on the standardized warning message.
6. Any other brand elements, like a logo or a slogan, may be displayed on the PDP. Brand elements cannot be metallic or fluorescent colours. The brand element must be equal to or smaller than the surface are of the standardized THC warning symbol on the PDP.
7. The PDP must display in regular standard san-serif black text on a white background the following information:
- Contact information of the license holder. Name, phone number, email
- Class of Cannabis
- Lot number
- Recommended storage conditions
- Packaging date
- Expiry date
- Net weight of product contents
- Net volume of product contents
- Number of discreet units, if applicable
- Net weight of discreet units, if applicable
- The warning statement: “KEEP OUT OF REACH OF CHILDREN / TENIR HORS DE LA PORTÉE DES ENFANTS” in upper case font
- Ingredients, if applicable. For requirements on how to list ingredients properly, see the Health Canada Cannabis Packaging and Labeling guidelines section 8.2 at the link: https://www.canada.ca/en/health-canada/services/cannabis-regulations-licensed-producers/packaging-labelling-guide-cannabis-products/guide.html
- Immediate small packaging must display these items on permitted peel-back or accordion style panel attached to the products primary packaging.
An example of a fully complaint package and labeling of a cannabis product with discreet units:


1. The standardized cannabis symbol
2. The brand name of the cannabis product
3. THC and CBD content
4. Health warning message
5. Other brand element
6. Other required information about the cannabis product
7. Non-required information about the cannabis product
8. Nutrition facts table
9. List of ingredients
10. Bar code
DISCLAIMER
This is a comprehensive summary of the Health Canada cannabis packaging and labeling guidelines, yet it is the responsibility of the company to ensure they are in compliance with the current and complete requirements for packaging and labeling their products. For the complete and current list of regulations, visit Health Canada’s list of rules pertaining to packaging and labeling of cannabis products. Also, for spacing and graphic design requirements such as spacing, colour, size requirements, and other information see website:
https://www.canada.ca/en/health-canada/services/cannabis-regulations-licensed-producers/packaging-labelling-guide-cannabis-products/guide.html